The first idea [1] for a glucose-responsive insulin (GRI, commonly referred to as smart insulin) was pitched almost 40 years ago, but to our knowledge only one compound has since undergone clinical evaluation. It seems a little surprising that it takes such a long time to develop a functioning GRI because its concept is actually relatively simple: At normal glucose concentrations, small amounts of insulin are released to keep blood glucose (BG) fairly constant. In response to rising glucose concentrations, for example after a meal, more insulin is released to limit the glucose rise and to return glucose concentrations back to normal. This closed-loop insulin release should limit BG variability and because insulin is only released when it is needed, it should also reduce the risk of over- and underdosing insulin. Two key elements are needed for a GRI: (1) A component that can ‘sense’ glucose and (2) a component that can respond and control the release of insulin. The concept may be simple, suggested solutions are complex and diverse [2].
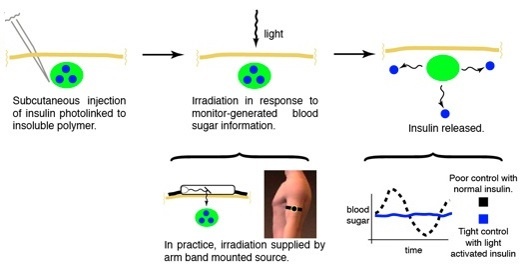 Figure 1: General concept of a light-responsive smart insulin [Source: The Friedman Lab]
Figure 1: General concept of a light-responsive smart insulin [Source: The Friedman Lab]
Matrix-based smart solutions
One type of GRI approaches embed insulin in (polymer) material, creating a physical depot of insulin in the body. In response to glucose, insulin is released from the depot either through a change of the matrix properties (e.g. degradation or a structural change) or through competitive glucose binding. The glucose sensing component can be an integral part of the matrix such as glucose oxidase in the transcutaneous smart insulin patch [3], or it can be a stand-alone component such as a continuous glucose monitoring (CGM) device. An interesting example of the latter comes from a research group at the University of Missouri that uses CGM data to trigger light-induced [4] insulin release from a subcutaneous depot. With this approach insulin molecules are bound to the matrix using a photocleavable linker, the matrix is injected and forms a subcutaneous depot. Above the depot, on top of the skin, a small (watch-sized) box is placed and emits light at a specific wavelength to break the linker and set the insulin free. The amount of light emitted depends on the sensor glucose level and a closed-loop insulin release platform has been established. In vitro and animal experiments have demonstrated proof-of-principle, but further optimisations are needed before the start of a clinical evaluation. A dual-hormone matrix, incorporating both insulin and glucagon, is a potential further development with the photoactivated technology. Two distinct linkers that each react to light at a specific wavelength enable the controlled release of either hormone and could add an extra layer of safety at low glucose concentrations.
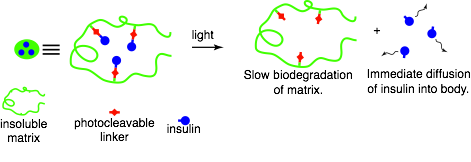 Figure 2: Detailed illustration of light-induced insulin release [Source: The Friedman Lab]
Figure 2: Detailed illustration of light-induced insulin release [Source: The Friedman Lab]
Engineering glucose responsiveness into insulin
In the manuscript [5] with this title, researchers from Merck & Co. describe their approach of modifying the insulin molecule itself to give it glucose-responsive properties, rather than creating a glucose-responsive matrix. Merck’s GRI that progressed into clinical trials, MK-2640, is an insulin analogue with conjugated carbohydrate groups that give the insulin an added ability to bind to the lectin receptor mannose receptor C-type 1 (MRC1). After binding to the insulin receptor MK-2640 induces a glucose lowering action and is subsequently degraded. Whereas after binding to MRC1, the insulin is degraded without insulin action. The affinity of MK-2640 to bind to MRC1 is greater than for the insulin receptor and the idea is that at normal glucose concentrations, most circulating insulin will be cleared from blood without insulin action and only a small amount will elicit a basal insulin action. Glucose competitively binds to the MRC1 receptor and will prevent insulin-MRC1 binding at rising and high glucose concentrations. A study [6] in dogs has shown that at high glucose concentration (280 mg/dL), circulating insulin levels were up by 30% compared to normal glucose concentration (80 mg/dL).
Clinical evaluation of a smart insulin
A milestone in GRI development has been reached recently with the first publication [7] of clinical trial data. Based on the results of the trial, Merck decided to discontinue the development of MK-2640, mainly due to an observed 25-fold lower potency compared to human insulin. Nevertheless, the trial has provided useful insights: At high glucose concentration, insulin clearance was not significantly lower (-6%) than at low glucose concentration, failing to show the kind of glucose-responsiveness observed in the dog study. Despite the lack of PK responsiveness, a 44% greater increase of the glucose infusion rate during the two-step clamp was seen compared to human insulin, showing for the first time a modest glucose-responsive effect of a smart insulin in humans. A result that the authors contribute mainly to hepatic insulin action. The trial furthermore identified an interesting limitation of this approach: saturation of MRC1 binding and subsequent insulin clearance was probably reached before insulin doses were tested that were high enough to elicit (substantial) insulin action. Meaning that at clinically relevant insulin doses, the glucose-responsiveness of MK-2640 was obviously limited by the MRC1 binding capacity. The good news is that Merck is not giving up on this approach and further GRI analogues with a higher affinity for the insulin receptor are being screened.
Hopefully more approaches will enter the clinic soon and accelerate GRI development. We look forward to keep track of all progress!




