Introduction
The glucose clamp is the gold standard for the determination of pharmacokinetic and pharmacodynamic effects of anti-diabetic drugs (e.g. insulins). During a typical glucose clamps the blood glucose (BG) lowering effect is antagonized by infusing glucose at a variable rate, so that BG is "clamped" at a pre-determined target level. In automated glucose clamps the glucose infusion rate is calculated automatically by a pre-specified algorithm.
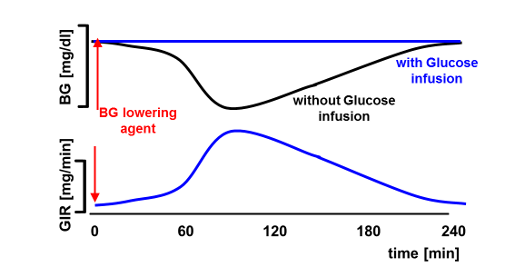
The first automated glucose clamp device was the Biostator, developed in the early 1970ies by Life Sciences Instruments, Elkhart, IN, USA, with an in-built algorithm for calculating GIRs [1]. While the Biostator was used in thousands of glucose clamp experiments, the algorithm had some limitations: The Biostator algorithm was designed in the 1970ies when fast-acting insulin analogues were not yet available. As there was no need for a fast adaptation of GIR, the algorithm reacts too slowly to compensate for the fast onset of action of these modern insulins leading to an initial substantial drop in BG at the time of the fastest rise in the insulin’s metabolic effect (“undershoot”) and thereafter a residual offset (“overshoot”) above target level when the metabolic action of the insulin declines after having reached maximum action.
We therefore developed a new clamp algorithm for rapid-acting insulins based on the PID (proportional–integral–derivative) controller approach. The new algorithm could be implemented in modern glucose clamp devices such as ClampArt and replace the slow reacting Biostator algorithm.
Methods
In-silico development
In the first development step the new Clamp-PID algorithm was designed and optimized. Based on our knowledge of pros and cons of the Biostator algorithm we designed the Clamp-PID algorithm using the PID-approach. The first part of the algorithm is proportional to the difference between the actual BG-value and the target level (p-part), the second part contains the mean GIR over the last minutes (i-part) and the last part takes the actual slope of the BG-values into account (d-part). The final GIR is a simple sum of these three parts including three unknown coefficients.
The aim of the following optimization of the algorithm was to find the best set of coefficients for the algorithm with respect to the actual set-up of the clamp experiment (particularly lagtime and precision of the glucose sensor) and the fast onset of modern rapid acting insulins. Simulated glucose clamps with differed coefficients were performed to find the best set of coefficients, i.e. to minimize the fluctuation in BG (precision) [2].
In-vitro verification

In the next development step the best set of coefficients was tested in-vitro using a 5L- container filled with glucose solution. The insulin action was created with appropriate infusion rates of water into the container which let the glucose solution overflow and thereby reduce glucose concentration. The glucose clamp quality parameters [2] precision (CV of BG values) and absolute control deviation (mean of absolute differences between target level and BG values) were compared between the new Clamp-PID- and the Biostator algorithm.
Validation in clinical study
Finally, the Clamp-PID algorithm was validated in a small clinical pilot study comparing the Clamp-PID- versus the Biostator algorithm in five healthy volunteers who received 0.3 U/kg of insulin aspart (NovoRapid, Novo Nordisk, Bagsvaerd, Denmark) under glucose clamp conditions on two different study days.
The ClampArt device was used on both days using either the Clamp-PID algorithm or the Biostator algorithm in random order. Pharmacodynamics were assessed for up to 12h after dosing, and reference BG values were obtained every 15 minutes. The BG target level was set to 81mg/dl (4.5mmol/l).
Results
In-silico development
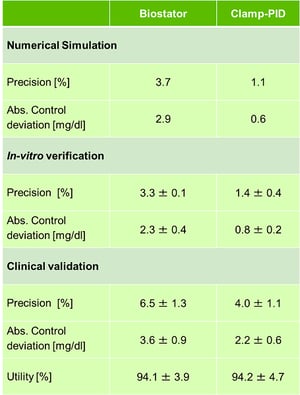
After thousands of simulated glucose clamps using different sets of coefficients the best set was found with a precision of 1.1% (see table). This precision is mainly driven by the precision of the simulated glucose sensor with only a very small contribution by the algorithm. Compared with the precision of simulated glucose clamps using the Biostator algorithm the Clamp-PID algorithm improves the overall precision of numerical glucose clamps more than 3-fold. As expected the BG curve is closer to target level for the Clamp-PID algorithm which is reflected by the almost 5-fold improvement of the absolute control deviation i.e. the sum of the absolute deviations of the BG values from target level.
In-vitro study
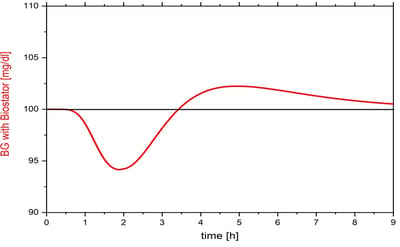
The in-vitro verification of the Clamp-PID algorithm with the optimal set of tuning parameters using the original ClampArt device confirmed the in-silico findings. The figure shows the BG-curve of an in-vitro automated glucose clamp using the Clamp-PID algorithm running on the ClampArt device (blue) with the best set of tuning parameters obtained from simulations in comparison with the BG-curve of the Biostator algorithm (red). Using the Biostator algorithm typically leads to a larger drop (“undershoot”) at the beginning of the glucose clamp where the insulin profile has its largest increase. The BG values obtained from the Biostator algorithm are about 8% below the target level of 100 mg/dl whereas the corresponding BG values with the Clamp-PID algorithm are only about 4% below target. After maximum insulin action is reached BG values with the Clamp-PID algorithm are exactly at target level (deviations of ± 1mg/dl are due to the precision of the real glucose sensor used in the ClampArt device) whereas BG-values with the Biostator algorithm are constantly about 3mg/dl above target until the end of insulin action. The shown BG curves are typical for glucose clamps with rapid acting insulins for the Biostator algorithm and the Clamp-PID algorithm, respectively.
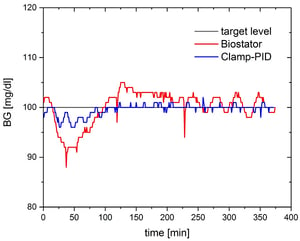
The clamp quality parameters were similar to the values obtained by numerical simulations and showed the same improvement for the Clamp-PID algorithm. Precision was improved by more than twofold down to 1.4% while absolute control deviation was improved by almost 3-fold down to 0.8mg/dl (see table).
Clinical study
The small clinical pilot study validated the in-vitro and in-silico results. Both precision and absolute control deviation were improved with the Clamp-PID algorithm versus the Biostator algorithm in all five paired glucose clamp experiments. Improvements were in the range of almost 40% for the two clamp quality parameters (see table). As expected, values for both precision and control deviation were higher in the clinical study than in the ideal environment of a lab or in numerical simulations as additional confounders such as blood clotting, movement of the subjects’ arm, impaired blood flow, etc. impair clamp quality. The utility was almost identical in both arms.
In summary, the higher clamp quality through the Clamp-PID algorithm predicted by the in-vitro study and the numerical simulations was confirmed in the clinical setting.
Conclusion and outlook
The new Clamp-PID algorithm substantially improved clamp quality versus the well-established Biostator algorithm in numerical simulations, in-vitro and in a clinical validation study.
The Clamp-PID algorithm will therefore be used for future automated glucose clamp studies with the ClampArt device.
Additional studies will investigate the suitability of the Clamp-PID algorithm for other settings such as insulin sensitivity clamps or PD investigations of slower-acting insulins.




