Why glucose clamps – and how?
The pharmacokinetic/pharmacodynamic (PK/PD) profiles of novel insulin formulations are commonly assessed by means of either automated or manual glucose clamps (watch the free online seminar to learn more). The metabolic effect of the investigated insulin is characterized by the glucose infusion rate (GIR) needed to keep blood glucose (BG) at a predefined target BG concentration during a hyperinsulinemic euglycemic clamp. In automated clamps BG is measured continuously and GIR is adjusted every minute – GIR adaptations in manual clamps, in contrast, “only” occur every 3-10 minutes. Because of the higher sampling rate the automated glucose clamp technique should have a better control quality compared to manual clamps. In addition, there is no room for bias caused by the clamper, which is theoretically possible with manual clamps due to GIR adjustments in anticipation of a certain PD effect [1].
Clamp.Art – a new dimension in automated glucose clamps
The new ClampArt device for automated glucose clamping, developed by Profil, Neuss, Germany, consists of two parts (see figure below): The central unit on the right side contains a computer with the ClampArt software including the algorithm which calculates the GIR needed to keep the blood glucose values at a predefined level. Furthermore the central unit includes all equipment for glucose measurements, i.e. the sensor unit and the pumps for blood sampling. The central unit is based on a mobile cart with a touchscreen monitor on top which serves as the user interface.
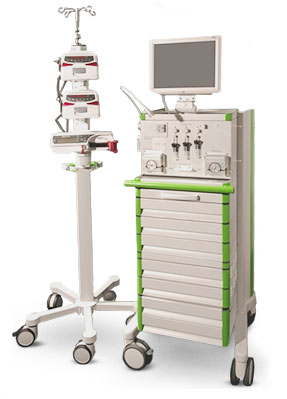
Figure 1: Clamp.Art, the innovative next generation automated glucose clamp device.
The second part of the ClampArt system consists of the infusion pumps at a remote docking station. The docking station is placed on the other side of the bed and is controlled by the central unit via cable connection.
One major improvement of the ClampArt device is the consideration of the blood dilution when determining the glucose concentration. This makes the measurement independent of fluctuations of the blood portion in the solution, which occurs at every automated clamp, for example due to motion artifacts of the patient.
Which parameters of clamp quality are available?
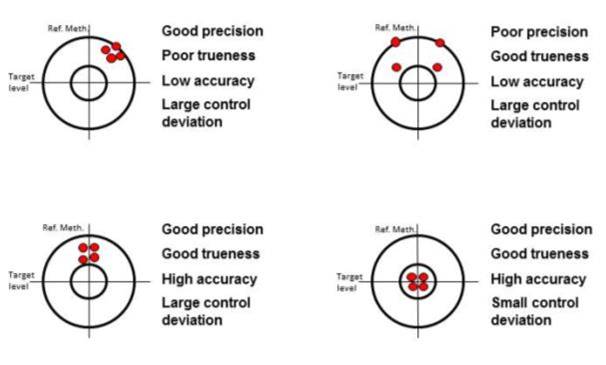
Figure 2: Profil Germany proposes glucose clamp quality parameters - Precision, Trueness, Accuracy, Control Deviation, Utility
Unfortunately the quality of glucose clamp procedures has hardly ever been published and there are no established parameters available. Profil, thus, has proposed, evaluated and just published [2] the following parameters to characterize clamp quality: trueness, precision, accuracy, control deviation (shown in the figure on the right) and utility (the percentage of time where the ClampArt device delivers blood glucose values and corresponding GIRs). The latter is of course only applicable to automated clamp devices. All these clamp quality parameters were assessed during a first series of clamps carried out with Clamp.Art and exhibited excellent results (for details see [2]).
What are the challenges for the future?
- A clear definition of “clamp quality” – which is also supported by regulatory authorities [1].
- A consensus of all clamp centres on how to measure clamp quality.
- The inclusion of these clamp quality parameters in future publications of every glucose clamp study.
Profil is determined to work towards achieving these next steps and welcomes anyone to discuss or aid in implementing such crucial quality control measures.




