The pandemic of the coronavirus disease (COVID-19) has rapidly changed our lives. Most facilities of daily life are closed, many people try to get accustomed to home-office and in many countries people are gated. Despite all these measures infection rates still rise exponentially in nearly all countries affected. In these times everyone should do their part in fighting the pandemic.
Profil, a CRO specialized on early phase clinical trials, is stepping up to this responsibility by offering support to companies developing vaccines against COVID-19. As one of the leading phase 1 units in Europe, we can offer experience with Europe's regulatory landscape as well as years of routine in conducting early phase trials in our two clinics in Germany. Combined with our team's previous experience working on novel vaccine developments this allows us to support companies looking for rapid and effective support in their clinical development.
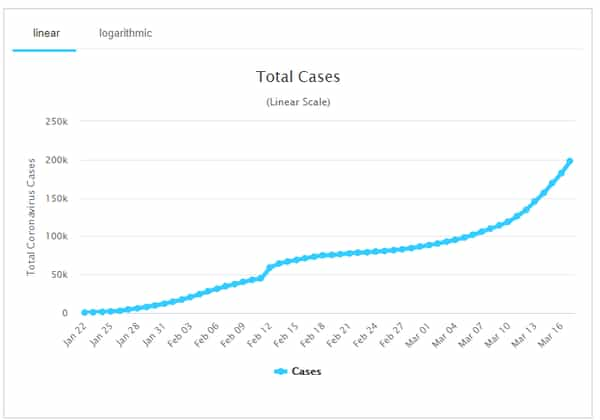
Total COVID-19 cases (Status 18 Mar 2020), Source: Worldometer - www.worldometers.info
Experts agree that vaccines against COVID-19 are urgently needed to stop the pandemic as soon as possible. Therefore, governments have released additional funds for research projects investigating, among others, vaccines for COVID-19.
In addition, authorities already indicated full support to facilitate and accelerate approval of new therapeutics and vaccines within the shortest possible timeframe.
Development of Vaccines: Pathways and Hurdles
In general, the development of new vaccines follows the usual path of novel compounds with three phases of clinical studies. Phase 1 trials investigating a candidate vaccine’s safety for the first time in human beings are usually done in a few dozen closely supervised healthy people. Phase 2 studies in some hundred patients provide first indications of efficacy followed by phase 3 trials which are mostly done as placebo-controlled trials in thousands, if not ten-thousands of subjects to adequately address the candidate vaccine's (surrogate) efficacy, reactogenicity (i.e., reactions occuring soon after vaccination as a physical manifestation of the inflammatory response), and safety.
 Image: https://www.istockphoto.com/de/portfolio/wavebreakmedia
Image: https://www.istockphoto.com/de/portfolio/wavebreakmedia
Without short-cuts the development of a COVID-19 vaccine would take years. Fortunately, the need for a faster development was already identified years ago, partly triggered by the devastating experience with the Ebola outbreak in West Africa where a 100% effective vaccine that had been under development for more than a decade still came too late to prevent the death of more than 11,000 people. This shortfall resulted, among others, in the foundation of the Coalition for Epidemic Preparedness Innovations (CEPI) whose mission it is to stimulate and accelerate the development of vaccines against emerging infectious diseases, e.g., through funding “new and innovative platform technologies with the potential to accelerate the development and manufacture of vaccines”.
Indeed, CEPI funds enabled the first pharmaceutical company to deliver a clinical batch in 42 days from sequence and to apply the first candidate vaccine against COVID-19 in the framework of a clinical study in the United States to a healthy woman on 16 March, 2020. Further developments are underway, so that more trials will hopefully start very soon.
FIH study with candidate COVID-19 vaccines: Fast and with high quality
If in an emergency situation like the current outbreak short-cuts can be found for the phase 2 and phase 3 studies of candidate vaccines against COVID-19, it will be even more important to do the initial phase 1 (first-in-human, FIH) study fast, but scrupulously.
This is anything but trivial as a lot of safety precautions have to be taken to mitigate risks for trial participants that are the first humans to receive a candidate vaccine. Participants in FIH-studies have experienced serious harm only on very rare occasions, nevertheless, the safety and well-being of trial participants should always be the utmost priority in clinical trials.
For this reason, the European Medicines Agency (EMA) and other competent authorities have published guidelines to mitigate and manage risks for trial participants in FIH-studies. Among many other strategies EMA have put emphasis on the suitability of investigator site facilities and personnel: “FIH/early clinical trials should take place in appropriate clinical facilities and be conducted by trained investigators and medical staff with appropriate levels of training and experience of early phase trials” (EMEA/CHMP/SWP/28367/07 Rev. 1, 20 July 2017, link).
At Profil, we have performed literally hundreds of early phase clinical trials including dozens of first-in-human studies. In our team, we have investigators with vast experience in vaccine studies, even though most of our studies at Profil investigated novel anti-diabetic agents. Nonetheless, there are important similarities to studies with potential candidate vaccines:
- Most of our studies were done with biological therapeutics, i.e., large and often complex proteins derived from living cells or animal tissues as are most vaccines. We are therefore familiar with the specifics of these drugs including potential side effects such as allergic reactions. We also have experience with nucleic acid therapeutics such as the first candidate for a COVID-19 vaccine that has been investigated in a clinical trial.
- While our set-up often allows investigating efficacy already in FIH-studies, the primary focus of all early phase clinical trials is safety. We therefore not only have equipment and appropriately qualified staff for acute emergency procedures as demanded in the EMA guidelines, our set-up ensures close and often continuous monitoring of vital functions, so that we can react quickly to potential adverse effects and prevent serious harm.
- Early phase clinical trials often pose challenges that are quite different to later phase studies (watch our free online seminar on FIH trial considerations). Complex procedures have to be done in a short time and documentation has to be meticulous to allow a highly standardized assessment of safety and efficacy of a new drug in a limited number of people. Enrolment of study participants has to be fast in order to prevent delays in subsequent trials. Profil has a high reputation for doing these kind of studies rapidly and according to highest standards. We have approximately 10,000 healthy volunteers in our subject database that are readily available for such studies. Several of our FIH-studies have successfully passed inspections by EMA and FDA which unerlines the high quality standards at Profil.
Profil’s motivation to support the development of COVID-19 vaccines
We feel that in the current emergency situation, everyone should do whatever possible to limit the already grim impact of the COVID-19 pandemic. We have therefore started to reach out to companies developing candidate vaccines against COVID-19 offering our help with testing these developments in FIH-studies at attractive conditions in healthy people. We are particularly committed to fighting against a further fast spread of COVID-19 as people with diabetes are reported to have an increased risk of developing the infection and a higher risk for a severe disease course and mortality. Thus, we feel that supporting the development of a COVID-19 vaccine will currently be the best way to achieve our vision of a better and healthier life for people with diabetes. Needless to say that our research for better anti-diabetic therapies and monitoring devices will continue at full steam.




